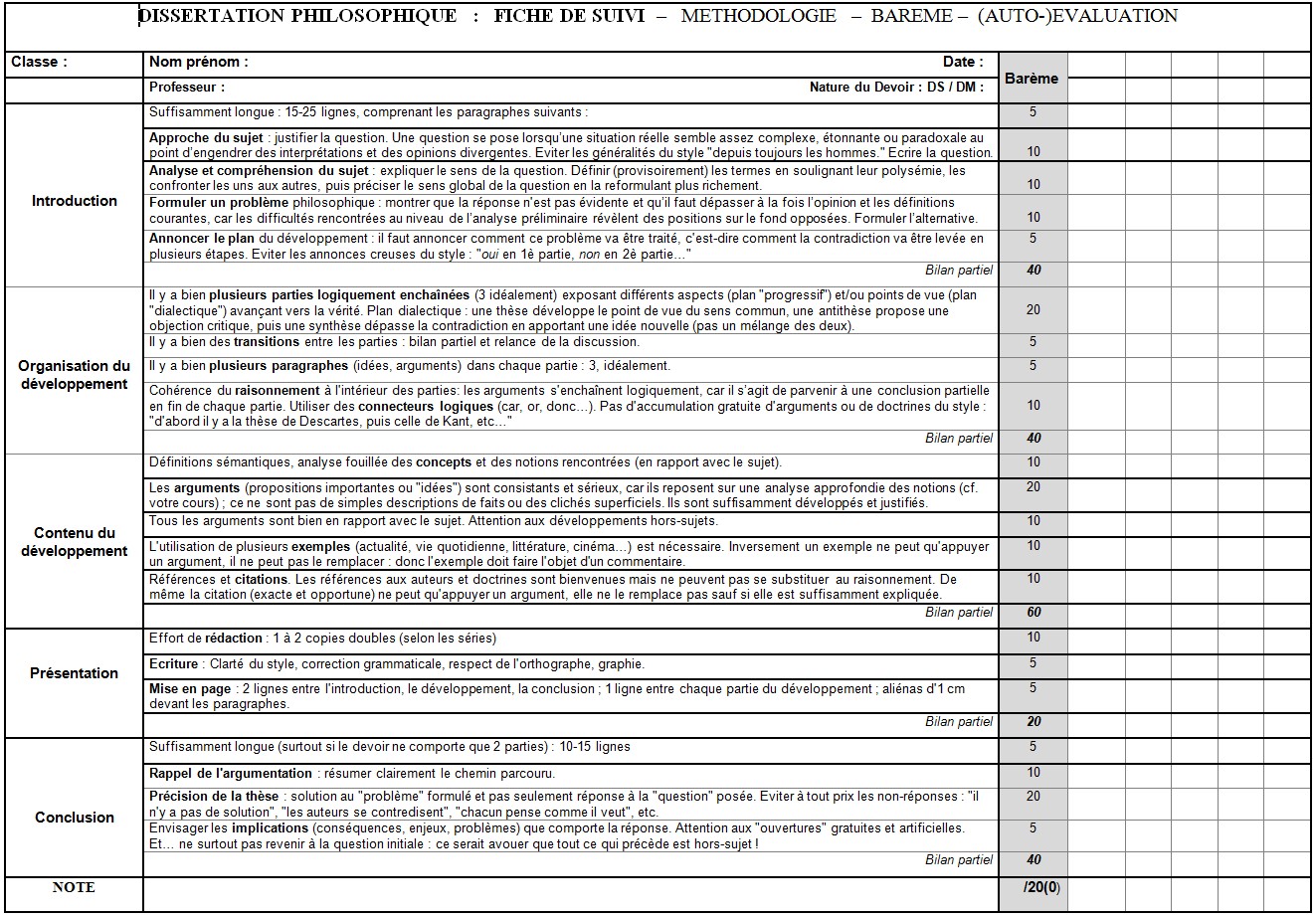
How to complete and balance an equation for reaction of.
Write the balanced chemical equation for this reaction. The reaction that occurs when an Alka-Seltzer tablet is dropped into a glass of water has sodium bicarbonate reacting with citric acid (H 3 C 6 H 5 O 7 ) to make carbon dioxide, water, and sodium citrate (Na 3 C 6 H 5 O 7 ).
The reason to write a chemical equation is to express what we believe is actually happening in a chemical reaction. One of the most useful applications of the concept of principal species is in writing net ionic equations.These are equations that focus on the principal substances and ions involved in a reaction--the principal species--ignoring those spectator ions that really don't get involved.
Write the balanced equation for the neutralization reaction between H3PO4 and NaOH in aqueous solution. Phases are optional. asked by Karen on May 14, 2014; chemistry. Complete and balance the molecular equation, including phases, for the reaction of aqueous ammonium bromide, NH4Br, and aqueous lead(II) acetate, Pb(C2H3O2).
Write balanced chemical equations for the following statements. i. NaOH solution is heated with zinc granules. ii. Excess of carbon dioxide gas is passed through lime water. iii. Dilute sulphuric acid reacts with sodium carbonate. iv. Egg shells are dropped in hydrochloric acid. v. Copper (II) oxide reacts with dilute hydrochloric acid.
By removing the spectator ion in this equation, we get the balanced ionic equation. Answer 3: The balanced net ionic equation will be, Balanced equations: Balanced equations are the equations in which the number of individual elements present on the reactant side must be equal to the number of individual elements present on the product side.
The chemical equation allows us to calculate the concentration of a solution of HCl by titration with the base NaOH (where the concentration of NaOH is accurately known). Let's suppose that our solution is 0.02500 L of an unknown concentration of the acid, HCl. We wish to find its concentration by titration with 0.1000 M NaOH.
Write the balanced molecular equation. Write the state (s, l, g, aq) for each substance. Split strong electrolytes into ions (the complete ionic equation). Cross out the spectator ions on both sides of complete ionic equation. Write the remaining substances as the net ionic equation. Note: charges in a net ionic equation are conserved.